INTRODUCTION
Rheumatoid arthritis (RA) is characterized by chronic systemic inflammation primarily affecting diarthrodial joints, resulting in disability and reduced quality of life as well as significant disease burden.1 Some recent studies have estimated the prevalence of RA in adults to be approximately 1.36 million in the United States (2014), 2.3 million in Europe (2017), and 1.24 million in Japan (2011).2–4
Currently, several treatment choices are available for RA including analgesics, nonsteroidal anti-inflammatory drugs (NSAIDs), steroids, conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), biological DMARDs (bDMARDs) (such as adalimumab [ADA], certolizumab [CZP], etanercept [ETN], golimumab [GOL], infliximab [IFX], and tocilizumab [TCZ]), and targeted synthetic disease-modifying antirheumatic drugs (tsDMARDs).5,6
As per the recent update of the European League Against Rheumatism (EULAR) RA management recommendations, methotrexate (MTX) (rapid escalation to 25 mg/wk) plus short-term glucocorticoids is recommended as the first strategy, aiming at >50% improvement within 3—and target attainment within 6—months.7 In the absence of poor prognostic factors, switching or including another csDMARDs (plus short-term glucocorticoids) is suggested.7 Any bDMARD or JAK inhibitor should be added to the csDMARD in the presence of poor prognostic factors. If this fails, any other bDMARD or tsDMARD is recommended.7
Since MTX is recommended as first-line therapy in RA in both the American College of Rheumatology (ACR) and EULAR guidelines,7,8 comparative efficacy analyses of biologics and traditional DMARDs in MTX-inadequate response (IR) patients might provide additional insights regarding the management of RA.9 Recent EULAR recommendations position JAK inhibitors as equal to bDMARDs; bDMARDs are no longer the preferred option for csDMARDs-IR patients who failed MTX, though the combination with MTX is preferred.7 Despite the progress achieved in the treatment of RA there are still unmet needs, as well as a need for improved application of currently available treatments—including better treatment options employing novel mechanisms of actions targeting new pathways.6 Recently, significant efforts have been undertaken in order to understand RA pathogenesis, including the role of JAKs in RA pathology.10,11
Reliable evidence on the comparative efficacy of the biologic antirheumatic agents is crucial for informing clinical and economic decisions about their optimal use. Only a few head-to-head trials of these therapies are available in the population of interest (eg, RA-BEAM12 showing the superiority of baricitinib (BARI) against adalimumab; ORAL STRATEGY,13 not showing noninferiority against adalimumab, both in the MTX-IR population). The objective of this network meta-analysis (NMA) was to evaluate the effectiveness of BARI 4 mg (oral, JAK 1/2 inhibitor) combined with MTX compared to other targeted synthetic/biologic DMARDs combination therapy with MTX, in moderate-to-severe RA MTX-IR patients. The aim is to provide evidence that will better inform health care providers’ decisions on patients’ treatment.
METHODS
Methods of Trial Selection, Quality Assessment, and Appraisal
Prior to the NMA, a systematic literature review (SLR) of randomized controlled trials (RCTs) of targeted synthetic/biologic DMARDs (abatacept [ABA], ADA, CZP, ETN, GOL, IFX, rituximab (RTX), TCZ, tofacitinib [TOFA], sarilumab [SARI], MTX, csDMARDs and BARI) in adult (≥18 years) moderate-to-severe RA MTX-IR patients was conducted (see Tables S1–S3 for population, intervention, comparator, outcomes and study type criteria used to identify studies). At the time the SLR was conducted, upadacitinib had not yet been given market authorization as a treatment for RA patients, and hence was not included. The SLR searches were conducted between 1999 and 2017, in MEDLINE, MEDLINE In-Process, EMBASE, Biosciences Information Service, the Cochrane Library, and Cochrane and European Union’s trials registers. Abstracts from EULAR, ACR, and the British Society for Rheumatology meetings were searched from 2013 to 2018. Only English-language publications were included, and no geographical restrictions were applied. Two reviewers screened the abstracts and full-text articles, while the data were extracted by one reviewer and validated by a second independent reviewer. Any uncertainties were resolved by a third reviewer. The search strategy and list of search terms can be found in the Supplementary Material methods (Tables S4–S7). The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used in development and reporting14 and quality assessment of trials was performed to standards recommended15 by the Centre for Reviews and Dissemination.16 A summary of the quality assessment of trials included in the NMA (primary analysis) is provided in the Supplementary Material (Table S8).
Endpoints and Time Points
Endpoints analyzed were ACR responses, defined by the ACR20, ACR50, and ACR70 response criteria.17 The ACR20 is a combined outcome defined as 20% improvement in the number of tender and swollen joints along with a 20% improvement in three of the following five criteria: patient global assessment, physician global assessment, functional ability measure (most frequently the Health Assessment Questionnaire (HAQ)), visual analogue pain scale, and erythrocyte sedimentation rate or C-reactive protein (CRP). ACR50 and ACR70 are defined as improvement levels of 50% and 70%, respectively, on the previously listed criteria.17 The time point of interest was 24 weeks, as this was the time point for the primary endpoint measures for newer biologic agents, including sarilumab.18 The 24-weeks’ time point was defined as trial visits scheduled at 24 weeks (± 4 weeks); trial visits from Week 20 to Week 28 were included.
Network Meta-Analyses (NMA)
NMAs were conducted using Bayesian mixed treatment comparisons as described in the National Institute for Health and Care Excellence (NICE) Decision Support Unit (DSU) Technical Support Documents (TSDs).15 ACR response was analyzed using logistic regression models with a binomial likelihood distribution. Both fixed-effect and random-effect simultaneous Bayesian models were fitted for the MTX-IR population. Model fit was assessed using the deviance information criterion (DIC) and residual deviance.19 The analysis incorporated the use of bDMARDs or tsDMARDs in combination with MTX where this was within license at 6 months, using the licensed doses for RA. For each analysis, a network diagram was drawn including the number of treatment arms contributing to each pairwise direct evidence. All results are presented here as odds ratios (ORs) with 95% credible intervals (CrI) of BARI 4 mg + MTX versus comparators, with ORs >1 indicating a better result for BARI 4 mg + MTX. A full description of the analysis methods and further results are provided in the Supplementary Material (Methodology, Results).
A series of preplanned sensitivity analyses (see the Supplementary Material, Table S10) were conducted—where possible or applicable—to evaluate the different assumptions made for the NMAs as well as the potential influence of treatment effect modifiers. These sensitivity analyses included the removal of trials (those limited to the Asian-Pacific region and/or using a low/previously unknown MTX dose, or open-label trials), addition of trials (up to 20% of prior bDMARDs), models fitting baseline and treatment effect separately, baseline risk adjustment, and frequentist NMAs.
Results
Trial Identification
The SLR identified 10 008 articles, of which 962 were deemed to be potentially relevant after title and abstract screening. After full-text screening, 147 trials with 322 publications were included in the review, which encompassed all RA patient populations. Of those, there were 39 trials that met the SLR inclusion criteria for the MTX-IR moderate-to-severe RA population, and which were eligible for the NMA. Of these, 10 trials allowed for up to 20% of patients with prior bDMARD use, who were only eligible for sensitivity analysis (1). Of the remaining 29 trials, 5 were excluded from the primary 24-week analysis because they were either 12-week trials (n = 3, EXXELERATE,20 REALISTIC,21 and Lan22) or were disconnected from the network due to switching or rerandomization prior to the 24-week time point (n = 2, Abe23 and CNTO 14824). Five additional trials were excluded as either all treatment arms were monotherapy (n = 3, ADACTA,25 SATORI,26 and MONARCH27) or because excluding monotherapy arms only left a single treatment arm (n = 2, ACT-RAY28,29 and JESMR30) (Supplementary Material, Figure S1). Furthermore, for the purpose of this analysis, treatment arms investigating monotherapy or treatment doses that were out of scope were excluded from trials with more than two treatment arms (eg, ORAL SCAN31 and ORAL STANDARD32 [TOFA 10 mg + MTX]; ORAL STRATEGY13 [TOFA 5 mg monotherapy]; Edwards33 [RTX 1000 mg monotherapy]; SERENE34 [RTX 500 mg + MTX]; and MOBILITY18 [SARI 150 mg + MTX]). The only BARI trial that was conducted in the MTX-IR population was RA-BEAM,12 in which only the 4 mg dose of BARI was investigated.
For the primary analysis, a total of 19 trials in the MTX-IR population with moderate-to-severe RA were included. Of these, four were conducted in the Asia Pacific region or only allowed for a low dose (<7.5 mg/wk) of MTX. For the sensitivity analysis allowing for trials with up to 20% of patients with prior bDMARD use, an additional 10 trials were eligible, increasing the total number of trials to 29. It is of note that all three TOFA trials (ORAL SCAN,31 ORAL STANDARD,32 and ORAL STRATEGY13) and both TCZ trials (LITHE,35 OPTION36) allowed for prior bDMARD use. Consequently, TOFA and TCZ could only be included into the NMA via the corresponding sensitivity analysis. With the exception of the ATTRACT37 trial (IFX 3 mg + MTX vs Placebo [PBO] + MTX), which only reported results for ACR20, all other trials presented results for all three ACR response rates. The common reference treatment is PBO + MTX. The full list of trials included in the primary NMA and sensitivity analyses, together with the endpoints available for the analysis at 24 weeks in each trial, is shown in the Supplementary Material (Table S9). The ACR response data as used in the analyses are provided in the Supplementary Material (Table S11).
Demographic Characteristics
Patient characteristics for each trial included in the NMA analysis conducted on the MTX-IR RA population are described in Table 1. In most trials included in the primary analysis, mean duration of disease was 7 to 11 years. Only one trial (AMPLE38) reported a mean duration of disease of less than 2 years. As was to be expected, the percentage of males was low (mostly 15 to 25%). Mean age (SD) ranged from 46.7 (12.2) to 57.20 (11.40). Across trials, the majority of patients were rheumatoid factor positive (>80%). The mean number of tender and swollen joints at baseline was influenced by corresponding inclusion criteria. Three trials reported baseline scores using the Clinical Disease Activity Index (CDAI) (mean: 36.0–38.6) and Simplified Disease Activity Index (SDAI) (mean: 21.2 –40.3). Most of the trials reported either DAS28–ESR or DAS28–CRP, and mean DAS28–ESR and DAS28–CRP scores ranged from 5.5–6.9 and 5.3–6.4, respectively.
Primary Analysis
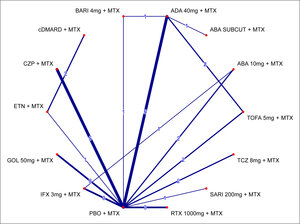
The network of evidence for the ACR20 response for the primary analysis is shown in Figure 1. The only difference between this network and those for ACR50/ACR70 is due to the ATTRACT37 trial (IFX 3 mg + MTX vs PBO + MTX), which only reported results for ACR20.
Both fixed- and random-effects models were run, and model fit showed similar results. However, in both models autocorrelation was present and random-effects models showed problems with convergence. Therefore, burn-in and thinning values were increased, and informative instead of noninformative priors were used. These measures considerably improved autocorrelation, but convergence of the random-effects models was still problematic. Therefore, fixed-effects models were chosen as the primary analysis approach and are presented here. As baseline demographics and clinical characteristics indicate some variability between trials (see Table 1), results for the random effects models are also shown (see the Supplementary Material, Tables S15–S17). In addition, Supplementary Material Table S27 provides model fit statistics as per the DIC and the overall residual deviance for the primary analyses and main sensitivity analyses.
BARI 4 mg + MTX was found to be statistically significantly more effective than ADA 40 mg + MTX (OR: 1.33; 95% Crl: 1.01–1.75); ABA 10 mg + MTX (OR: 1.45; 95% CrI: 1.01– 2.10); IFX 3 mg + MTX (OR: 1.63; 95% CrI: 1.16–2.30); and RTX 1000 mg + MTX (OR: 1.63; 95% CrI: 1.08–2.46) on the ACR20 response. No differences were found between BARI 4 mg + MTX and comparators on the ACR50 response. On the ACR70 response, BARI 4 mg + MTX was found to be statistically significantly more effective than ADA 40 mg + MTX (OR: 1.37; 95% CrI: 1.02–1.87); ABA 10 mg + MTX (OR: 1.86; 95% CrI: 1.09–3.17); and RTX 1000 mg + MTX (OR: 2.26; 95% CrI: 1.11–4.47) (Figure 2). Results for all pairwise comparisons are shown in the Supplementary Material (Tables S12–S14).
Sensitivity Analyses
Baseline Risk Adjustment. Bayesian network meta-regressions adjusting for the variability in PBO response rates across trials were conducted. Scatter plots of PBO response rates (ie, baseline risk) against the treatment effects for each trial showed that increasing PBO responses were associated with a decreasing treatment effect for each of the three outcomes (see the Supplementary Material, Figure S2 [A]-[C]).
Some differences were seen in the sensitivity analysis adjusting for baseline risk, where BARI 4 mg + MTX was shown to be statistically significantly more effective compared to a higher number of comparators as found in the primary analysis. This was mainly because of higher point estimates or smaller CrIs (for ACR20, the additional comparator was GOL 50 mg + MTX; for ACR50, additional comparators were GOL 50 mg + MTX, IFX 3 mg + MTX, ABA 10 mg + MTX, and RTX 1000 mg + MTX) (Figure 3).
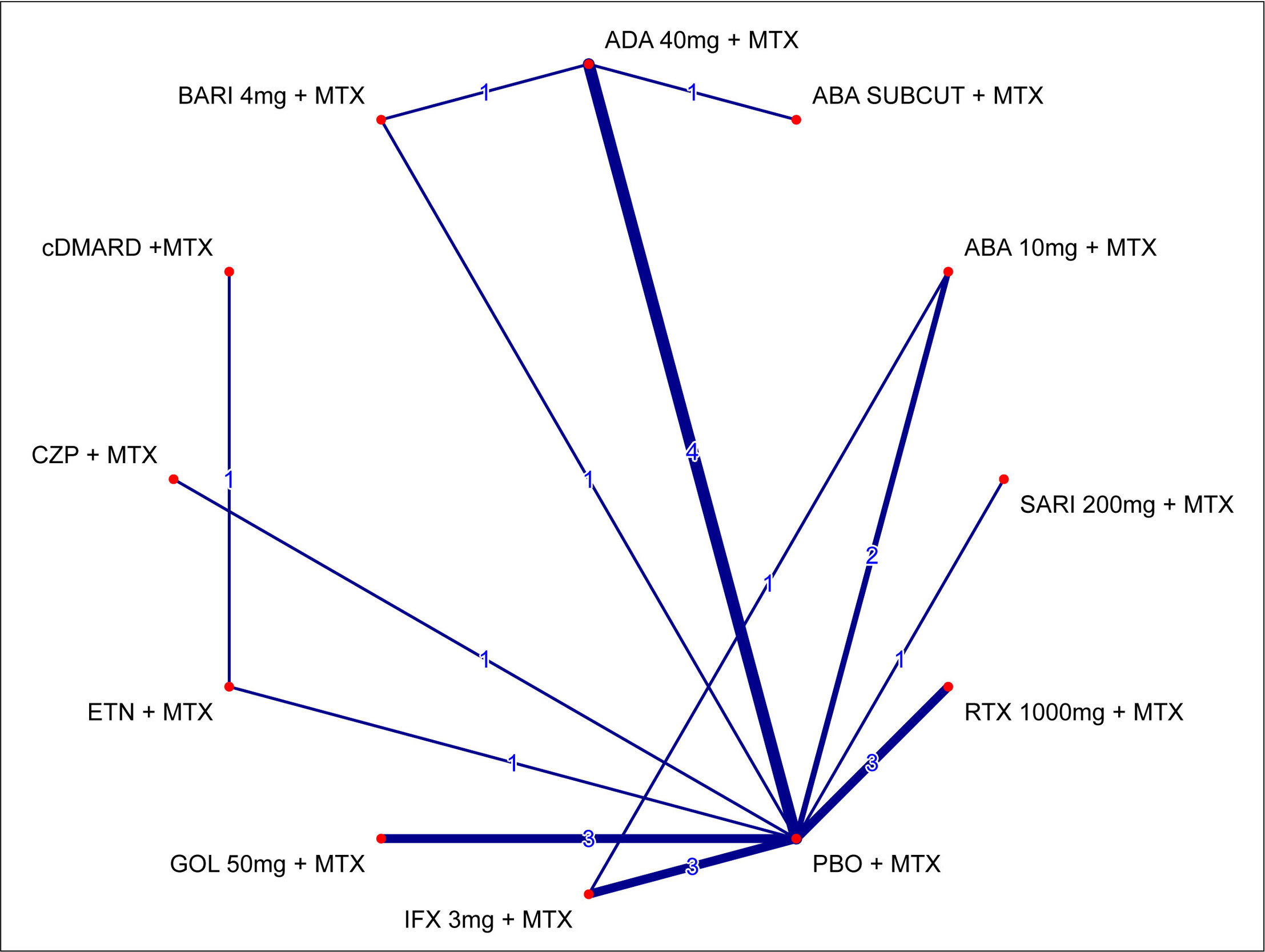
Inclusion of Trials in Which up to 20% of Patients had Prior bDMARD Use. Although up to 20% of patients with prior bDMARDs are often allowed in clinical trials for csDMARDs/MTX-IR populations, those patients might constitute a source of bias, mainly in head-to-head comparisons versus TNFs, as they have previously failed the same mode of action, and responses using a second TNF might be expected to be lower. Consequently, these trials were excluded from the primary analysis, but explored via a prespecified sensitivity analysis that followed the NICE Technology appraisal guidance TA37557 and its associated assessment report.58 This sensitivity analysis included 10 additional trials with up to 20% of patients with prior bDMARD use (J-RAPID52, Kang,53 RAPID1,55 RAPID2,56 LITHE,35 OPTION,36 ORAL SCAN,31 ORAL STANDARD,32 ORAL STRATEGY,13 and RACAT54), and allowed comparisons versus TOFA 5 mg + MTX and TCZ 8 mg + MTX. It is of note that patients in these trials had a shorter mean duration of disease, a lower percentage of being rheumatoid factor positive, and slightly higher mean DAS28-ESR or DAS28-CRP scores at baseline. The network of evidence for the ACR20 response for this sensitivity analysis is presented in Figure 4. As for the primary analysis, the only difference between networks comes from the ATTRACT37 trial.
No major changes from the primary model were observed for ACR20, ACR50, and ACR70 responses, but for the fact that BARI 4mg + MTX was no longer statistically significantly more effective than ABA 10 mg + MTX for the ACR20 response (OR: 1.42; 95% CrI 0.98–2.04) (Figure 5). This sensitivity analysis allowed the comparisons versus TOFA 5 mg + MTX and TCZ 8 mg + MTX. No statistically significant differences were observed for BARI 4 mg + MTX versus TOFA 5 mg + MTX, whereas BARI 4 mg + MTX was statistically significantly more effective than TCZ 8 mg + MTX on ACR20 (Figure 5). Results for all pairwise comparisons are shown in the Supplementary Material (Tables S21–S23).
Trials Conducted in the Asia Pacific Region / Using Low Dose MTX. This sensitivity analysis excluded trials if they were only conducted in Asian-Pacific countries or if the trials had included low (<7.5mg/wk) or unknown MTX doses. It addresses the potential impact on treatment response due to low dose background MTX use, as those patients might not be truly MTX failures. Trials allowing for lower doses of MTX as an inclusion criterion might therefore constitute a source of heterogeneity, and these are predominantly conducted in Asian-Pacific countries. This sensitivity analysis followed the NICE Technology appraisal guidance TA375.57 Trials excluded were GO-FORTH,42,59 Kim,45 Li,60 and RAPID-C, all conducted in Asian-Pacific countries.61 It is of note that patients in these trials reported lower mean numbers of tender joint count (TJC) and swollen joint count (SJC), primarily due to lower corresponding inclusion criteria. As a result, 15 trials were included for ACR20 and 14 trials for ACR50 and ACR70. Specifically, for ADA 40 mg, one out of three trials was excluded,45 two out of three trials were excluded for GOL 50 mg (GO-FORTH and Li49), and CZP was no longer part of this sensitivity analysis, in addition to TCZ 8 mg and TOFA 5 mg. No changes from the primary model were observed for ACR20, ACR50, and ACR70 responses (see the Supplementary Material, Section 10.3 for more details).
Further Sensitivity Analyses. Apart from the sensitivity analyses described previously in greater detail, additional sensitivity analyses (excluding open-label trials, models fitting baseline and treatment effect separately, and frequentist NMAs) were conducted to assess the robustness of the primary models in this MTX-IR population. The sensitivity analysis excluding open-label trials compensated for the potential introduction of bias. However, this only excluded one ETN trial.47 Results for all these sensitivity analyses were consistent with the direction and magnitude of the primary results.
To illustrate the different results across the primary and sensitivity analyses, estimated median ACR response rates, plus corresponding 95%-CrIs, are available for each of the treatments (see Supplementary Material Section 12, Figures S4–S16).
While the investigation of inconsistency through node splitting had been preplanned (see the Supplementary Material, Table S10) it was not performed as there were only two closed loops coming from one trial, respectively (ATTEST41, RA-BEAM12).
Discussion
The treatment goal in RA is to control inflammation, relieve pain, and reduce disability associated with the condition.62 ACR and EULAR guidelines recommend MTX as the first-line therapy for RA. However, many patients do not experience adequate or complete responses with MTX,8 creating the need for alternative treatment options in patients who are either refractory or intolerant to MTX. It is important to determine the efficacy of optimal treatment options for these patients; this analysis provides a comprehensive comparison of the efficacy of BARI 4 mg treatment in combination with MTX versus alternative targeted synthetic/biologic DMARDs combination therapy with MTX in moderate-to-severe RA MTX-IR patients. Sarilumab (SARI), which has been approved recently, has also been added to the list of comparators. This NMA followed best practice guidelines, using Bayesian mixed treatment comparisons, allowing simultaneous comparisons of BARI with all treatment options. All available literature was considered. Extensive predefined sensitivity analyses were conducted to test the different assumptions made for the NMAs and to assess the robustness of results.
Primary analyses were conducted for ACR response (20%, 50%, and 70% improvement) at the 24-week time point. As was to be expected, results showed that all active comparators (bDMARDs/tsDMARDs) in combination with MTX were more effective than PBO + MTX. The combination of BARI 4 mg and MTX was either superior or equally effective across all ACR responses, with the ORs favoring BARI in the majority of cases. The results are in line with other meta-analyses conducted to evaluate the efficacy of BARI in RA patients with inadequate response to csDMARDs including MTX.63,64 Sensitivity analyses showed consistent results with the primary analysis in both direction and magnitude.
It is noteworthy that two of the three ETN trials were against csDMARD + MTX (ie, not the main common comparator PBO + MTX), and one of them was the only open-label trial in this NMA.47 Furthermore, the analysis method in this open-label trial used last-observation carried forward (LOCF) imputation, which differs from the nonresponder imputation used in the other trials. Consequently, ACR response rates in this open-label trial were higher than in any other trials in this NMA. Furthermore, TOFA 5 mg + MTX and TCZ 8 mg + MTX could only be analyzed via the sensitivity analysis allowing for trials with up to 20% of prior bDMARD use. This must be taken into account when interpreting the results.
Potential limitations to this analysis include the time span over which trials were conducted (1999 to 2017), during which patient characteristics and treatment approaches may have changed. As a result, ACR responses might alter over time and result in a “placebo creep.”65 Although methods such as the baseline risk adjustment aim to address this problem, it cannot be fully solved by analytical methods alone.
As the analyses focused on the MTX-IR population, the only BARI trial conducted in this population was RA-BEAM,12 in which only the 4 mg dose of BARI was investigated. Therefore, no conclusions could be drawn for BARI 2 mg. Of note, the 4 mg dose of BARI constitutes the most commonly approved dose within this population.66,67
Advantages of NMAs are the ability to combine information from different clinical trials for which direct (head-to-head) evidence may not be available. The focus on the MTX-IR population and therapies in combination with MTX reduced variability. The clinical effectiveness evidence was drawn from a comprehensive and systematic review of RCTs undertaken to assess treatments for RA patients and is, therefore, of general applicability for MTX-IR populations. Results for all sensitivity analyses were consistent with the direction and magnitude of the primary results, providing a robust basis of evidence.
In a very competitive market, BARI, a once-daily oral drug with a rapid onset of action and similar or better comparative effectiveness to other bDMARDs on the market, could be a preferred option for treating RA patients instead of TNF inhibitors. The results of this NMA are supported by the recently published 2019 EULAR guidelines regarding the position of tsDMARDs in the treatment algorithm of RA patients so that no preference is given to bDMARDs over the tsDMARD class.7
In conclusion, the results of the NMA suggest that BARI 4 mg + MTX is an efficacious treatment option in the MTX-IR population as evidenced by the robustness of results and differences favoring BARI.
Funding Sources
This study was conducted by Eli Lilly and Company Ltd. Eli Lilly and Company Ltd. provided funding for the research and employees of Eli Lilly and Company Ltd. contributed to the study’s protocol design, data interpretation, data analysis, and manuscript development and review.
Acknowledgments
Initial conceptual work for NMA and consulting was provided by Alan Brnabic (Eli Lilly and Company, West Ryde, NSW, Australia), Mark Belger (Eli Lilly and Company, Lilly Research Centre, Windlesham, U.K.), and Zbigniew Kadziola (Eli Lilly Regional Operations GmbH, Vienna, Austria). Medical writing support was provided by Samprati Avasthi, an employee of Eli Lilly and Company.


_-_(mtx-ir_population__primary_analysis).jpg)
_(mtx-ir_population__sensitivity_analysis___1__).jpg)
_(mtx-ir_population__sensitivity_analysis___2__).jpg)
_-_(mtx-ir_population__primary_analysis).jpg)
_(mtx-ir_population__sensitivity_analysis___1__).jpg)
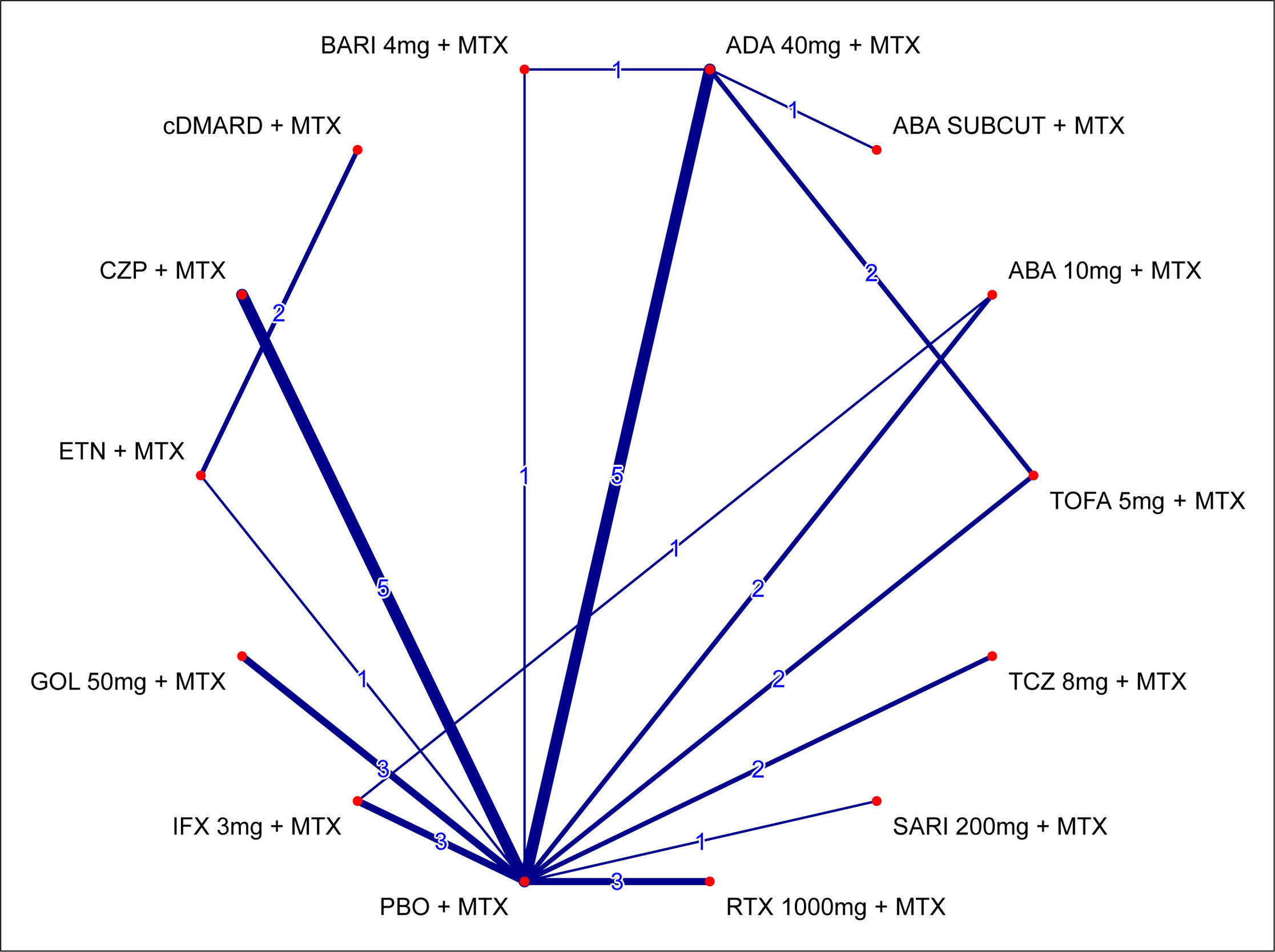
_(mtx-ir_population__sensitivity_analysis___2__).jpg)